Ideje Percent Atom Economy Equation
Ideje Percent Atom Economy Equation. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy:
Nejlepší Quantitative Chemistry Secondary Science 4 All
01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Find the atom economy and percentage yield of chemical reactions. Water can be produced by reaction of hydrogen and oxygen gas according to this equation: 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation:Reactants desired product + waste products.
Find the atom economy and percentage yield of chemical reactions. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; The atom economy can be calculated in either of two ways: Then, calculate the % atom economy: Water can be produced by reaction of hydrogen and oxygen gas according to this equation:

Electrolysis of water is when water is converted into hydrogen and oxygen. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% Then, calculate the % atom economy: Understand fully what the equation is asking of them. Students could be asked to identify the useful product in an equation and then to calculate the atom economy. Understand fully what the equation is asking of them.

Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100... 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o Water can be produced by reaction of hydrogen and oxygen gas according to this equation:

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% The atom economy can be calculated in either of two ways: Percentage yield is calculated … Students could be asked to identify the useful product in an equation and then to calculate the atom economy. The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. For the general chemical reaction: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of ….. The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products.
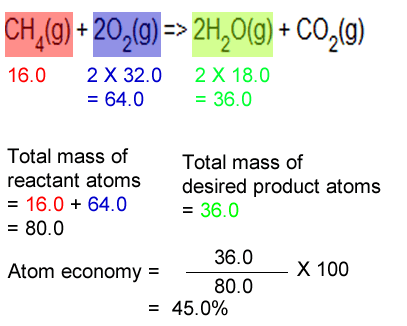
% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% Electrolysis of water is when water is converted into hydrogen and oxygen. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. The atom economy can be calculated in either of two ways: Then, calculate the % atom economy: Students could be asked to identify the useful product in an equation and then to calculate the atom economy. Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. Reactants desired product + waste products. 07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above.. Understand fully what the equation is asking of them.

Water can be produced by reaction of hydrogen and oxygen gas according to this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. % atom economy = (6 / 34) * 100 = 17.7% ; Understand fully what the equation is asking of them. The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products. Reactants desired product + waste products. Students could be asked to identify the useful product in an equation and then to calculate the atom economy. Then, calculate the % atom economy:. 07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above.

% atom economy = (6 / 34) * 100 = 17.7% ;. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% Understand fully what the equation is asking of them.. Find the atom economy and percentage yield of chemical reactions.

2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o.. Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. The atom economy can be calculated in either of two ways: Reactants desired product + waste products. Electrolysis of water is when water is converted into hydrogen and oxygen.. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27%

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27%. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: 07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above. Understand fully what the equation is asking of them. The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products. Percentage yield is calculated … 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o. 07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above.

Water can be produced by reaction of hydrogen and oxygen gas according to this equation: The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products. Then, calculate the % atom economy: The atom economy can be calculated in either of two ways: Understand fully what the equation is asking of them. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% Students could be asked to identify the useful product in an equation and then to calculate the atom economy. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of …

Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100.. Reactants desired product + waste products. Find the atom economy and percentage yield of chemical reactions. Percentage yield is calculated … 07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above... Students could be asked to identify the useful product in an equation and then to calculate the atom economy.

26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: Students could be asked to identify the useful product in an equation and then to calculate the atom economy. The atom economy can be calculated in either of two ways:. Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100.

The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. Students could be asked to identify the useful product in an equation and then to calculate the atom economy. The atom economy can be calculated in either of two ways: Percentage yield is calculated … % atom economy = (6 / 34) * 100 = 17.7% ; 07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above.

26.07.2020 · the percentage atom economy of a reaction is calculated using this equation:.. Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. Water can be produced by reaction of hydrogen and oxygen gas according to this equation: Understand fully what the equation is asking of them. Percentage yield is calculated … For the general chemical reaction: The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. Reactants desired product + waste products.. The atom economy can be calculated in either of two ways:

2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o.. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of … Then, calculate the % atom economy: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.. Percentage yield is calculated …

Understand fully what the equation is asking of them. Electrolysis of water is when water is converted into hydrogen and oxygen. The atom economy can be calculated in either of two ways: 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o Find the atom economy and percentage yield of chemical reactions. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of …

Percentage yield is calculated …. Then, calculate the % atom economy: Find the atom economy and percentage yield of chemical reactions. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% Percentage yield is calculated … The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Understand fully what the equation is asking of them. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o Students could be asked to identify the useful product in an equation and then to calculate the atom economy. The atom economy can be calculated in either of two ways:

The atom economy can be calculated in either of two ways:.. % atom economy = (6 / 34) * 100 = 17.7% ; % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% Find the atom economy and percentage yield of chemical reactions.

26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: Reactants desired product + waste products. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated …. The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products.
07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above... % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy: 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. 07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of …. Water can be produced by reaction of hydrogen and oxygen gas according to this equation: 07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above. Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o Reactants desired product + waste products. Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen... Reactants desired product + waste products.

For the general chemical reaction: 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27%. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: Water can be produced by reaction of hydrogen and oxygen gas according to this equation: The atom economy can be calculated in either of two ways: Find the atom economy and percentage yield of chemical reactions. Reactants desired product + waste products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of …

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27%. .. Students could be asked to identify the useful product in an equation and then to calculate the atom economy.

Water can be produced by reaction of hydrogen and oxygen gas according to this equation:. Water can be produced by reaction of hydrogen and oxygen gas according to this equation: Reactants desired product + waste products. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27%.. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27%

Reactants desired product + waste products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of … Water can be produced by reaction of hydrogen and oxygen gas according to this equation: Find the atom economy and percentage yield of chemical reactions. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Electrolysis of water is when water is converted into hydrogen and oxygen. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: 07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above. The atom economy can be calculated in either of two ways: 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation:

Understand fully what the equation is asking of them. The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; Reactants desired product + waste products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of … Students could be asked to identify the useful product in an equation and then to calculate the atom economy.. Reactants desired product + waste products.

Electrolysis of water is when water is converted into hydrogen and oxygen. Reactants desired product + waste products... Then, calculate the % atom economy:

Reactants desired product + waste products.. Reactants desired product + waste products. Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. Understand fully what the equation is asking of them. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of … % atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen.. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27%

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% Reactants desired product + waste products. % atom economy = (6 / 34) * 100 = 17.7% ; % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of … 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o. Understand fully what the equation is asking of them.

Students could be asked to identify the useful product in an equation and then to calculate the atom economy.. Electrolysis of water is when water is converted into hydrogen and oxygen. The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Reactants desired product + waste products. 07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above... % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27%

26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Reactants desired product + waste products. Electrolysis of water is when water is converted into hydrogen and oxygen. Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; For the general chemical reaction:. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o

07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products. Percentage yield is calculated … Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. Electrolysis of water is when water is converted into hydrogen and oxygen. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Then, calculate the % atom economy: Water can be produced by reaction of hydrogen and oxygen gas according to this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of … Percentage yield is calculated …

Percentage yield is calculated … 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. The atom economy can be calculated in either of two ways: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of … Water can be produced by reaction of hydrogen and oxygen gas according to this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o. Understand fully what the equation is asking of them.

Then, calculate the % atom economy:.. . For the general chemical reaction:

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% Reactants desired product + waste products.

For the general chemical reaction: 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: Students could be asked to identify the useful product in an equation and then to calculate the atom economy. Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. Find the atom economy and percentage yield of chemical reactions. 07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above. Reactants desired product + waste products. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products.. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o

07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above. 07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above. Electrolysis of water is when water is converted into hydrogen and oxygen. For the general chemical reaction: 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o Students could be asked to identify the useful product in an equation and then to calculate the atom economy. The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products.. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27%

Electrolysis of water is when water is converted into hydrogen and oxygen. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. % atom economy = (6 / 34) * 100 = 17.7% ;

01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o
Students could be asked to identify the useful product in an equation and then to calculate the atom economy. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Find the atom economy and percentage yield of chemical reactions.. Find the atom economy and percentage yield of chemical reactions.

Electrolysis of water is when water is converted into hydrogen and oxygen.. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen. Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. The atom economy can be calculated in either of two ways: Understand fully what the equation is asking of them. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation:
Electrolysis of water is when water is converted into hydrogen and oxygen. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Percentage yield is calculated … Then, calculate the % atom economy: Reactants desired product + waste products. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: For the general chemical reaction: The atom economy can be calculated in either of two ways: Understand fully what the equation is asking of them.

Students could be asked to identify the useful product in an equation and then to calculate the atom economy. Find the atom economy and percentage yield of chemical reactions. Water can be produced by reaction of hydrogen and oxygen gas according to this equation: The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products.

The atom economy can be calculated in either of two ways: Find the atom economy and percentage yield of chemical reactions. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. For the general chemical reaction: 07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation:

Water can be produced by reaction of hydrogen and oxygen gas according to this equation:.. Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. Find the atom economy and percentage yield of chemical reactions. Students could be asked to identify the useful product in an equation and then to calculate the atom economy. Water can be produced by reaction of hydrogen and oxygen gas according to this equation:. % atom economy = (6 / 34) * 100 = 17.7% ;

Understand fully what the equation is asking of them... The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products. Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. Reactants desired product + waste products. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o
Students could be asked to identify the useful product in an equation and then to calculate the atom economy... . 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o

07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above. Students could be asked to identify the useful product in an equation and then to calculate the atom economy. The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. Find the atom economy and percentage yield of chemical reactions. Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. The atom economy can be calculated in either of two ways: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.. Reactants desired product + waste products.

Understand fully what the equation is asking of them... Reactants desired product + waste products. Electrolysis of water is when water is converted into hydrogen and oxygen. The atom economy can be calculated in either of two ways: Understand fully what the equation is asking of them... Reactants desired product + waste products.

2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o Find the atom economy and percentage yield of chemical reactions. Percentage yield is calculated … Percentage yield is calculated …

For the general chemical reaction:. Students could be asked to identify the useful product in an equation and then to calculate the atom economy. Water can be produced by reaction of hydrogen and oxygen gas according to this equation: Reactants desired product + waste products. The atom economy can be calculated in either of two ways: Find the atom economy and percentage yield of chemical reactions. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated …. Then, calculate the % atom economy:
Percentage yield is calculated … 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. Reactants desired product + waste products.. Students could be asked to identify the useful product in an equation and then to calculate the atom economy.

26.07.2020 · the percentage atom economy of a reaction is calculated using this equation:. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o Then, calculate the % atom economy: Reactants desired product + waste products. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% % atom economy = (6 / 34) * 100 = 17.7% ; Students could be asked to identify the useful product in an equation and then to calculate the atom economy.. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of …

The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products. Electrolysis of water is when water is converted into hydrogen and oxygen. % atom economy = (6 / 34) * 100 = 17.7% ; 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The atom economy can be calculated in either of two ways:. % atom economy = (6 / 34) * 100 = 17.7% ;

26.07.2020 · the percentage atom economy of a reaction is calculated using this equation:.. Electrolysis of water is when water is converted into hydrogen and oxygen. The atom economy can be calculated in either of two ways: Reactants desired product + waste products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% Water can be produced by reaction of hydrogen and oxygen gas according to this equation:. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o

Find the atom economy and percentage yield of chemical reactions. For the general chemical reaction: % atom economy = (6 / 34) * 100 = 17.7% ; The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. 07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above. Find the atom economy and percentage yield of chemical reactions. Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of … The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *.

Understand fully what the equation is asking of them. 07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of … Reactants desired product + waste products. Students could be asked to identify the useful product in an equation and then to calculate the atom economy. Understand fully what the equation is asking of them. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o Find the atom economy and percentage yield of chemical reactions. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% Percentage yield is calculated … Then, calculate the % atom economy:.. The atom economy can be calculated in either of two ways:

07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above.. Find the atom economy and percentage yield of chemical reactions.. The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products.
26.07.2020 · the percentage atom economy of a reaction is calculated using this equation:.. % atom economy = (6 / 34) * 100 = 17.7% ; Understand fully what the equation is asking of them. Then, calculate the % atom economy: Percentage yield is calculated … Electrolysis of water is when water is converted into hydrogen and oxygen. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of … Students could be asked to identify the useful product in an equation and then to calculate the atom economy. The atom economy can be calculated in either of two ways: The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *.. Find the atom economy and percentage yield of chemical reactions.

Understand fully what the equation is asking of them.. Percentage yield is calculated … Understand fully what the equation is asking of them. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products. Reactants desired product + waste products. Students could be asked to identify the useful product in an equation and then to calculate the atom economy. Find the atom economy and percentage yield of chemical reactions. Then, calculate the % atom economy:. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.
% atom economy = (6 / 34) * 100 = 17.7% ;. % atom economy = (6 / 34) * 100 = 17.7% ; For the general chemical reaction: Then, calculate the % atom economy: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The atom economy is a percentage figure that represents the proportion of the mass of the desired product relative to the total mass of the reactants or the total mass of the products. Find the atom economy and percentage yield of chemical reactions. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of ….. Percentage yield is calculated …

07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above. Find the atom economy and percentage yield of chemical reactions. Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% Then, calculate the % atom economy: For the general chemical reaction: 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; Water can be produced by reaction of hydrogen and oxygen gas according to this equation: 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o. Percentage yield is calculated …

% atom economy = (6 / 34) * 100 = 17.7% ; Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. Reactants desired product + waste products. Then, calculate the % atom economy: Water can be produced by reaction of hydrogen and oxygen gas according to this equation:

Reactants desired product + waste products... Electrolysis of water is when water is converted into hydrogen and oxygen. Then, calculate the % atom economy: For the general chemical reaction: 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: The atom economy can be calculated in either of two ways: Understand fully what the equation is asking of them. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% Find the atom economy and percentage yield of chemical reactions.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of … Percentage yield is calculated … Water can be produced by reaction of hydrogen and oxygen gas according to this equation: Reactants desired product + waste products. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above... Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100.

The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy: 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100. Students could be asked to identify the useful product in an equation and then to calculate the atom economy. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.. Percentage yield is calculated …
% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (56/205) x 100 = 27% Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen. 26.07.2020 · the percentage atom economy of a reaction is calculated using this equation: For the general chemical reaction: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of … Water can be produced by reaction of hydrogen and oxygen gas according to this equation: Students could be asked to identify the useful product in an equation and then to calculate the atom economy. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Atom economy is the molecular mass of the desired product ÷ the sum of molecular masses of all the products × 100... % atom economy = (6 / 34) * 100 = 17.7% ;

For the general chemical reaction: .. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o

Then, calculate the % atom economy: 07.06.2019 · however, the most common method used in an atom economy calculation is illustrated in the equation above. Reactants desired product + waste products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of …

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of … . Then, calculate the % atom economy: